Jun 28 2017
The chemical industry is now adapting and finding new renewable sources to sustain its activities due to the uncertain future of oil feedstocks and environmental pressure.
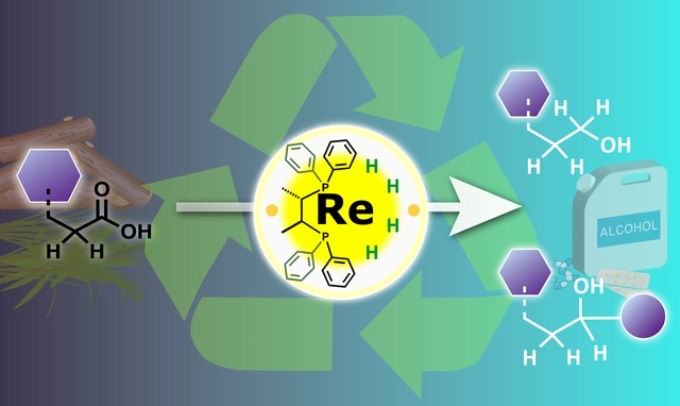 High valence rhenium used to recycle organic waste for a carbon neutral society. A rhenium catalyst in a high oxidation state is used to hydrogenate carboxylic acids present in organic waste, producing a range of useful alcohol products. © Nagoya University
High valence rhenium used to recycle organic waste for a carbon neutral society. A rhenium catalyst in a high oxidation state is used to hydrogenate carboxylic acids present in organic waste, producing a range of useful alcohol products. © Nagoya University
Biomass from sources including agricultural waste, wood and even human garbage signifies an extensively available renewable feedstock that has yet to be completely tapped. The issue is that most biomass is a mixture of different chemicals, which are difficult to separate and use in high-value products such as pharmaceuticals and plastics.
Carboxylic acids are very common chemical groups in biomass and their chemical reactions are mainly difficult to regulate in these mixtures. Presently used "catalytic hydrogenation" with metal catalysts can convert the acids into more beneficial alcohol groups, but also adds to the complexity of the biomass because of catalyst decomposition and other side reactions.
A team of Researchers from Nagoya University recognized the need for more selective transformations of carboxylic acid groups and so examined a different chemical method to the catalysis of biomass.
Traditionally, low-valent transition metal complexes are used for hydrogenation of carboxylic acids. But we found better selectivity under milder conditions using a high-valent complex, which also attacked carbon-hydrogen bonds next to the carboxylic acid.
Masayuki Naruto, Lead Author
Hydrogenation is fundamentally a reduction, during which the metal catalyzes relocation of electrons to the carboxylic acid. Low-valence metals are electron rich, which make them the clear choice for hydrogenation of carboxylic acids. However, the team demonstrated that high valence metals could also react with the carboxylic acids by a different pathway, which provided a lot more control over the reactivity.
The idea that high-valent transition metals are effective for this kind of reaction might go against traditional wisdom, but we have shown the potential of this approach for making high-value chemical products from biomass. Although, the rhenium metal catalyst we used here is rather expensive, we are now looking at recycling the catalyst and alternative tungsten and molybdenum catalysts, which should make this a truly economically viable approach for getting useful products from biomass in the future.
Susumu Saito, Team Leader