The BreakThrough Analyzer (BTA) is a high-performance, flexible gas delivery and management system for precise characterization of adsorbent performance.
- Excellent design reduces dead volume and produces precise experimental outcomes
- Configurable with up to six precise mass flow controls and two vapor sources
- Patented high-performance blending valves
- Sample activation up to 1050 ℃
- The thermostated environmental chamber enables uniform temperature control, even while employing vapors
- Easily links to commercial Mass Spectrometers (MS) and Fourier Transform Infrared Analyzers (FTIR)
- Secure door lock system improves operator safety
Description
Engineered for Performance
The BreakThrough Analyzer (BTA) is a versatile gas supply and management device for precisely determining adsorbent performance under process-relevant circumstances. It uses a flow-through method for accurate gas/vapor combination adsorption data.
An extremely well-optimized, safe apparatus for gathering equilibrium and transient adsorption data for systems with several components. The BTA offers unmatched versatility in experimental design and can be built with up to six precise mass flow controllers and proprietary high-performance blending valves.
With the least amount of dead volume possible, the excellent gas-delivery architecture guarantees accurate control over composition and flow rate.
The premium stainless-steel column has a 0.05 to 2.5 grams of adsorbent capacity. The accurate, durable, and dependable resistance furnace allows for automated sample activation up to 1050 °C.
Operating pressures may be adjusted from air to thirty bar using a servo-positioned controlled valve. Cold spots are eliminated by the thermostated environmental chamber, which provides consistent temperature control throughout the system up to 200 °C. During the analysis, operator safety is guaranteed by the BTA secure door lock system.
The BTA can add vapor generators so crucial probe molecules, like water, can be used in experiments. The BTA is easily interfaced with mass spectrometers and Fourier transform infrared systems sold commercially to identify and quantify gases.
Safety and Features
An extremely well-optimized, secure tool for gathering equilibrium and transient adsorption data for systems with several components.

Image Credit: Micromeritics Instrument Corporation
- Optional connection to shut off system
- Optional valve to shut off all MFC’s
- Optional gas detector in the thermostated environmental chamber
- Optional valve shut-off valve
- Shut off from the software
- Door closed during the analysis
- Furnace temperature control alarm
- Thermostated environmental chamber temperature control alarm
Specifications
Source: Micromeritics Instrument Corporation
| . |
. |
| Furnace Temp Max |
1050 °C |
| Thermostated environmental chamber Temp Max |
200 °C |
| Sample Mass |
Up to 2.5 g |
| Sample Volume |
Up to 2.5 mL |
Additional Specs
- Optimized for research-scale sample sizes with interchangeable reactor beds
- Ultra-low dead volume for rapid signal response
- Automated switching between purge and process gases
- Configurations for gas-vapor and vapor-vapor separation
- The door stays locked while the analysis is being conducted to safeguard both the user and the analysis from changing temperatures
- Touch Screen
- Patented “No Dead Volume” mixing valve with rapid switching
- Determination of breakthrough curves
- Study of kinetic performance of adsorbents
- Study of co-adsorption and displacement
- Determination of sorption selectivity
- High-resolution separations using small sample quantities
- Dynamic adsorption and desorption experiments
- Determination of single- and multi-component adsorption data
- In-situ sample preparation up to 450 °C with a SS column and 1050 °C with a quartz column
- Completely automated control via PC
- Up to six high-precision mass flow controllers
- Programmable total pressure, flow rate, composition, and temperature
Configurations
Mass Spectrometer (MS)
A mass spectrometer (MS) is frequently used for multicomponent adsorption experiments to track the residual gas composition. The most used detection system for breakthrough analysis is the MS.
FTIR Analyzer (FTIR)
FTIR spectrometers are frequently chosen for cutting-edge experimental research like the separation of xylenes or other aromatic compounds.
Humidity Sensor
Directly tracks water content at a reasonable cost. It can be valuable, particularly in production control applications.
Sample Preparation System
Small amounts of active substance can be combined with an inert carrier to create a homogenous sample and increase analytical repeatability.
CO2 Sensor
Facilitates inexpensive, direct tracking of CO2 content. It could be helpful, particularly in situations involving production control.
Sample Column (Different Volume)
The BTA can be utilized with a range of column diameters to handle extrudates, pellets, and powders, among other sample morphologies.
MFC and Mixing Valves (Maximum 6 Gas Inlets)
To improve analytical capabilities and broaden the range of experiments that can be carried out, the BTA could be equipped with more mass flow controls and blending valves.
Vapor Source (Max of 2)
The BTA’s alternative vapor sources are compatible with moisture and other vapors, such as xylenes and other aromatics.
Application and Materials
Popular Applications
Direct Air Capture

Image Credit: Micromeritics Instrument Corporation
Due to low air concentrations of carbon dioxide and other contaminants, such as moisture, DAC is challenging. To offset carbon emissions, captured CO2 can be sold, stored underground, or transformed into value-added chemicals.
CO2 Adsorption
Image Credit: Micromeritics Instrument Corporation
Significant point sources of carbon dioxide emissions include chemical plants, power plants, and refineries. Compared to direct air capture, these facilities frequently need to operate under different circumstances due to the greater quantities of carbon dioxide.
Olefin/Paraffin Separations

Image Credit: Micromeritics Instrument Corporation
They are essential to the petrochemical sector and are employed in the energy-intensive, higher-CO2 separation processes that produce polymers like polyethylene and polypropylene.
Natural Gas Separation

Image Credit: Micromeritics Instrument Corporation
Before being used in homes and businesses for cooking and heating, natural gas—a combination of hydrocarbons and other gases—must be cleaned.
Toxic Gas Adsorption
Image Credit: Micromeritics Instrument Corporation
In addition to being developed to capture harmful gases such as sulfur dioxide, hydrogen sulfide, and nitrogen dioxide from natural gas or other process inputs, porous materials are utilized for personal protection.
Water Adsorption
Image Credit: Micromeritics Instrument Corporation
Since freshwater supplies are scarce in many regions of the world due to desert climates or rising water use for agriculture, air harvesting technologies could prove essential for keeping these regions clean.
Materials
Zeolites
Image Credit: Micromeritics Instrument Corporation
Zeolite 5A, 13X, or LiX, which have a high selectivity for adsorbing nitrogen, are employed in the commercial sector for pressure swing adsorption to separate air and produce oxygen.
Silicas

Image Credit: Micromeritics Instrument Corporation
Amine-functionalized silicas are utilized for CO2 direct air capture (DAC) and are efficient and highly selective adsorbents.
Porous Membranes/Monoliths

Image Credit: Micromeritics Instrument Corporation
Zeolites covered with porous membranes and monoliths, or MOFs, are frequently employed to increase the operating efficiency of separation operations.
Activated Carbon

Image Credit: Micromeritics Instrument Corporation
Car fuel systems release volatile organic compounds (VOCs), which are reduced by activating carbon canisters that absorb and retain the VOCs.
Porous Aluminas

Image Credit: Micromeritics Instrument Corporation
Effective adsorbents with potential uses for CO2 extraction from natural gas are alumina-supported ionic liquids.
Metal-Organic Frameworks

Image Credit: Micromeritics Instrument Corporation
Highly selective adsorbents, MOFs work well for demanding commercial applications such as DAC, CO2, and CH4, olefins and alkynes, and alkanes and olefins.
Breakthrough Adsorption Theory
Image Credit: Micromeritics Instrument Corporation
A useful method for figuring out an adsorbent’s sorption capacity under flow circumstances is breakthrough analysis. Using dynamic breakthrough adsorption instead of static adsorption measurements has several benefits.
- Gather multicomponent adsorption data with ease.
- Determine adsorbate selectivity
- Replicate process conditions
Sample preparation is a crucial stage in the analysis process while doing breakthrough analysis to avoid pressure drop and mass transfer constraints. When there is insufficient interstitial space between particles to support the gas flow rate, pressure loss occurs.
Mass transfer is limited when a material’s pore size is comparable to the adsorbate’s kinetic diameter. For optimal outcomes, it is consequently essential to size the particles appropriately.
Multicomponent Vapor Analysis
Up to two vapor streams can pass through the packed column of the Micromeritics BTA at once. The thermostated environmental chamber keeps all gases and vapors inside the instrument at a consistent temperature, which also keeps these vapor streams from condensing during analysis.
A bubbler produces vapor streams, enabling a carrier gas to saturate with the desired vapor. Measurements of multicomponent ethanol/water breakthrough on zeolite 13× are shown in the figure below.
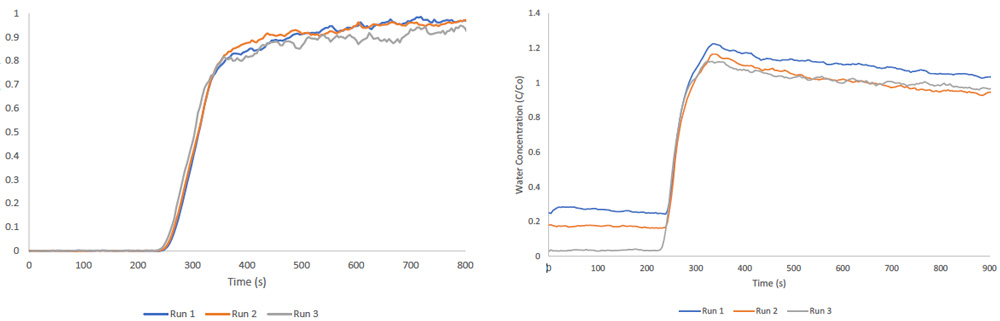
Image Credit: Micromeritics Instrument Corporation
Examining a Breakthrough Curve
- Complete Adsorption
At the breakthrough column’s exit, there is no trace of the fully adsorbed adsorbate gas.
- Breakthrough
First, the breakthrough column’s exit detects the presence of the adsorbate gas. Although the adsorbent can no longer completely adsorb the gas entering the breakthrough column, the gas still adsorbs
- Saturation
The adsorbate gas can now easily travel through the column since the adsorbent has attained saturation and cannot absorb it any longer.
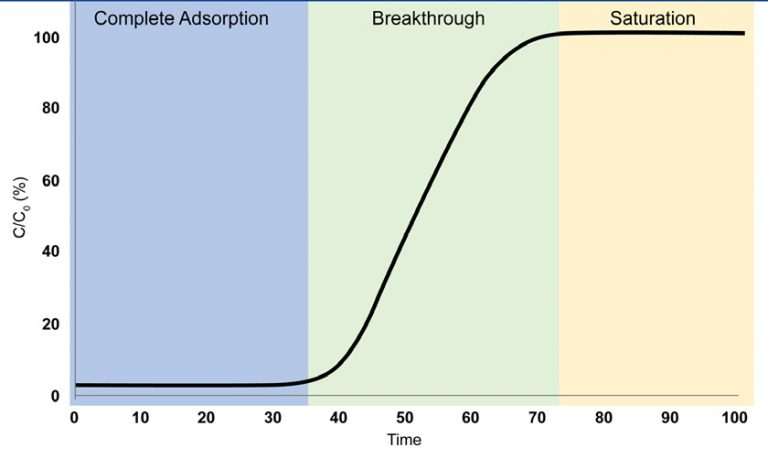
Image Credit: Micromeritics Instrument Corporation
Carbon Dioxide Adsorption
Adsorption studies for single-component carbon dioxide breakthrough were carried out on zeolites 13× and 5A, as well as metal-organic frameworks MIL-53(Al) and Fe-BTC. All samples were subjected to an equimolar gas stream, including 10 sccm carbon dioxide and 10 sccm nitrogen at 30 °C.
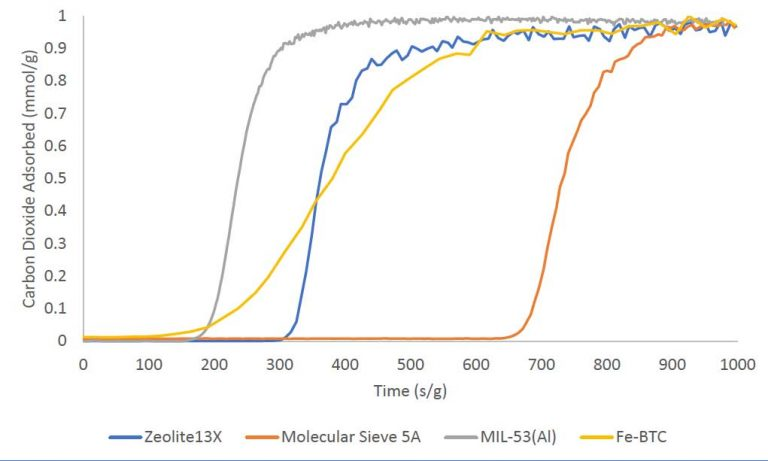
Image Credit: Micromeritics Instrument Corporation
To help pinpoint the beginning of the groundbreaking experiment, a tracer gas of 1 sccm of helium was also mixed into the input gas stream. The four materials’ breakthrough curves are presented below on a mass-normalized axis. The overall amount of CO2 adsorbed is distributed as follows: MIL-53(Al) > Fe-BTC > Zeolite 13× > Molecular Sieve 5A. The total amount adsorbed in mmol/g is displayed in the table below.
Source: Micromeritics Instrument Corporation
| Material |
Carbon Dioxide Adsorbed (MMOL/G) |
| Zeolite 13X |
2.94 |
| Molecular Sieve 5A |
3.52 |
| MIL-53 (AI) |
1.23 |
| FE-BTC |
2.30 |
High-Pressure Adsorption
Zeolite 13× has been thoroughly researched for use in adsorption and catalysis. Zeolite 13× was employed as an adsorbent in this investigation to gather breakthrough curves for carbon dioxide adsorption at pressures ranging from 1 to 10 bar. The equimolar flow rates of 10 sccm carbon dioxide and 10 sccm nitrogen were used to acquire these measurements.
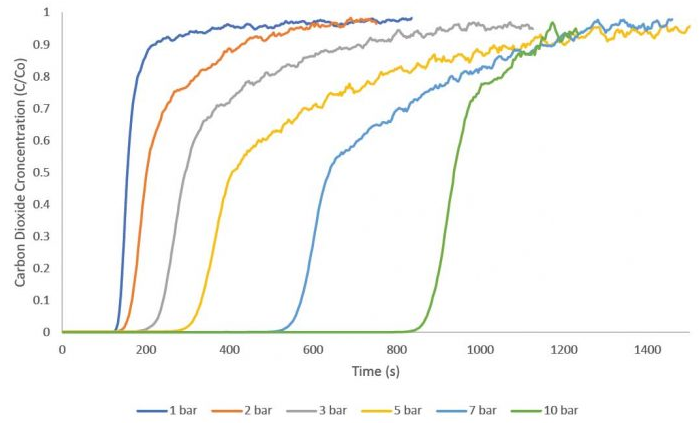
Image Credit: Micromeritics Instrument Corporation
The groundbreaking experiment was initiated using a 1 sccm stream of helium as a tracer gas. All measurements were taken at a temperature of 30 °C for analysis. The zeolite 13× sample was reactivated overnight between each test to guarantee total carbon dioxide desorption. The image illustrates how increasing pressure causes the breakthrough time to grow consistently throughout subsequent tests.
An equilibrium adsorption amount was determined for each curve by calculating the breakthrough equation after carbon dioxide breakthrough readings. The amount of carbon dioxide adsorbed at 1, 2, 3, 5, 7, and 10 bar total pressure was then displayed on an isotherm that was built. Zeolite 13× absorbed around 15 mmol/g of carbon dioxide at 10 bar.
Although breakthrough isothermal data cannot be directly associated with static adsorption measurements, it may be used to evaluate an adsorbent under circumstances relevant to the process.
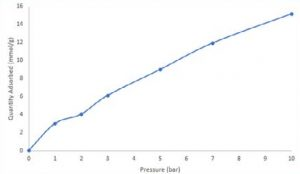
Image Credit: Micromeritics Instrument Corporation
Software
BTA Software
Image Credit: Micromeritics Instrument Corporation
The most user-friendly, adaptable, and thorough analytic program for adsorption experiments is MicroActive.
The program is extremely versatile, user-friendly, and simple, enabling various experimental circumstances and automating processes such as sample activation and analysis. It also supports cyclic experiments.
The BTA system solves the breakthrough equation for the most challenging samples, precisely and consistently characterizes adsorbents, and analyzes data using extensive analytical techniques when paired with the industry-leading MicroActive analysis software.
MicroActive Software enables users:
- Data reduction from MS
- Quantity adsorbed selectivity