Jun 19 2019
Photocatalysts that absorb light energy and use it to separate water into oxygen and hydrogen gain considerable scholarly interest because of the influence of hydrogen as a promising clean energy source.
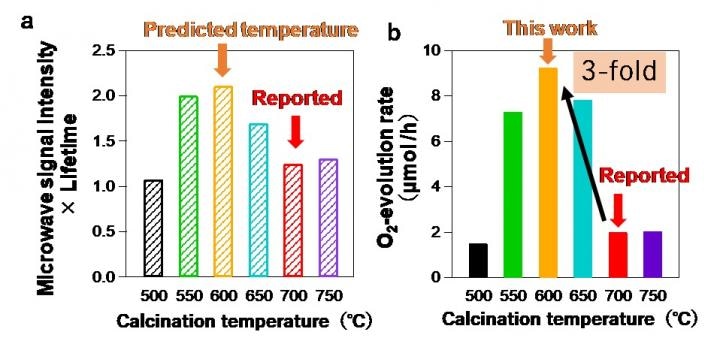 (a) Product of microwave photoconductivity intensity and lifetime of oxyhalide photocatalyt 2 vs calcination temperature. The microwave measurements suggest the optimal temperature of 600 °C that is 100 °C lower than the reported one. (b) O2-evolution rate of oxyhalide photocatalyt 2 prepared by different calcination temperature. (Image credit: Osaka University)
(a) Product of microwave photoconductivity intensity and lifetime of oxyhalide photocatalyt 2 vs calcination temperature. The microwave measurements suggest the optimal temperature of 600 °C that is 100 °C lower than the reported one. (b) O2-evolution rate of oxyhalide photocatalyt 2 prepared by different calcination temperature. (Image credit: Osaka University)
However, the optimization of photocatalyst candidate materials generally demands a significant time investment. Currently, scientists at Osaka University have shown a relationship between easy-to-measure quantities and catalyst performance that could offer a quick evaluation technique.
Although the conversion of light energy to chemical energy with the help of photocatalysts has been extensively described, the continual optimization of photocatalytic materials is important for their successful application.
The activity of photocatalysts is influenced by their properties such as crystallinity, surface area, and different electronic features. The methods and specific conditions employed to prepare them can have an impact on these properties, resulting in a wide variety of materials that could be assessed.
Experiment setup and test of every generated material is a prolonged stage in the development process that is not yet accelerated so far. In a report published in ACS Energy Letters, Osaka scientists have demonstrated the link between time-resolved microwave conductivity (TRMC) measurements and the photocatalytic performance of semiconductor materials. TRMC is a simplistic process that enables photocatalysts to be assessed in powder form, which results in considerably higher throughput.
We have been able to show that the oxygen evolution rate of a photocatalyst—which is a measure of activity—can be determined from the photoconductivity and the half-lifetime determined by TRMC. Applying this relationship to materials makes evaluating their potential much more efficient.
Hajime Suzuki, Study Lead Author, Osaka University
The scientists used the study outcomes to compute the optimum processing temperature for PbBiO2Cl, a material that had not been investigated much. They were able to yield an analog that had an evident quantum efficiency of 3%, which is three times higher than what had been realized in prior studies with higher processing temperatures.
We hope that the principles of our findings can be widely applied to improve the efficiency and ease of screening materials, finding candidates, and choosing synthesis conditions. In terms of the broader picture, high throughput processes could accelerate the development of cleaner energy solutions.
Akinori Saeki, Study Corresponding Author, Osaka University