May 5 2020
Researchers at Rice University have developed an efficient and inexpensive device that breaks down water to generate hydrogen fuel.
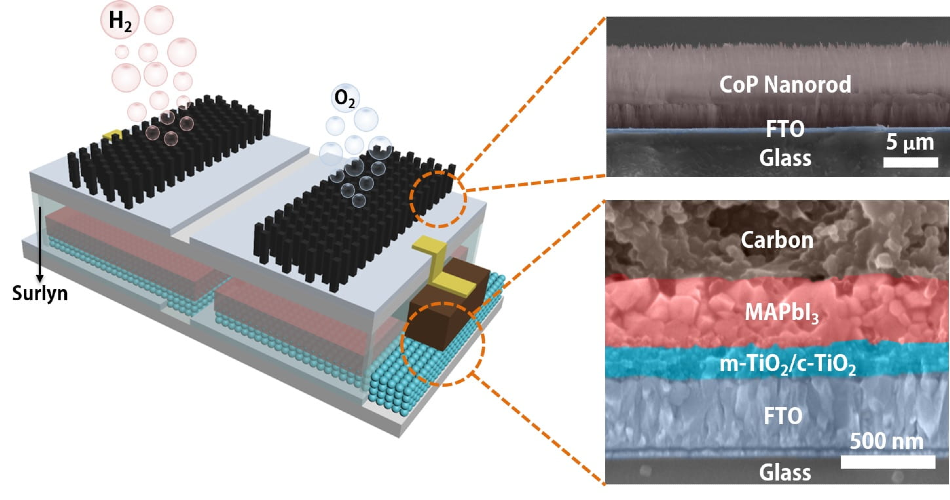 A schematic and electron microscope cross-section show the structure of an integrated, solar-powered catalyst to split water into hydrogen fuel and oxygen. The module developed at Rice University can be immersed into water directly to produce fuel when exposed to sunlight. Image Credit: Illustration by Jia Liang.
A schematic and electron microscope cross-section show the structure of an integrated, solar-powered catalyst to split water into hydrogen fuel and oxygen. The module developed at Rice University can be immersed into water directly to produce fuel when exposed to sunlight. Image Credit: Illustration by Jia Liang.
The Brown School of Engineering lab of Jun Lou, a Rice materials scientist, has designed this platform, which combines perovskite solar cells and catalytic electrodes that generate electricity when activated by sunlight. When currents flow to the catalysts, they break down water into oxygen and hydrogen, at a sunlight-to-hydrogen efficiency as high as 6.7%.
Although catalysis of this kind is very common, the researchers integrated the electrodes and a perovskite layer into a single module that generates hydrogen with no further input when it is immersed in water and placed in sunlight.
The platform launched by Lou, lead author and Rice postdoctoral fellow Jia Liang together with their collaborators in the American Chemical Society journal ACS Nano is a self-sustaining synthesizer of fuel that must be easy to mass-produce.
The concept is broadly similar to an artificial leaf. What we have is an integrated module that turns sunlight into electricity that drives an electrochemical reaction. It utilizes water and sunlight to get chemical fuels.
Jun Lou, Professor of Materials Science and Nanoengineering, Rice University
Perovskites are crystals that have cube-like lattices with the ability to tap light. To date, the most efficient perovskite solar cells realize an efficiency of more than 25%, but the materials are costly and get stressed by heat, humidity, and light.
Jia has replaced the more expensive components, like platinum, in perovskite solar cells with alternatives like carbon. That lowers the entry barrier for commercial adoption. Integrated devices like this are promising because they create a system that is sustainable. This does not require any external power to keep the module running.
Jun Lou, Professor of Materials Science and Nanoengineering, Rice University
According to Liang, the perovskite may not be the main component, but the polymer surrounding it, which safeguards the module and enables it to be submerged for longer periods.
Lou said, “Others have developed catalytic systems that connect the solar cell outside the water to immersed electrodes with a wire. We simplify the system by encapsulating the perovskite layer with a Surlyn (polymer) film.”
Liang added, the patterned film enables sunlight to reach the solar cell while safeguarding it and acts as an insulator between the electrodes and the cells.
With a clever system design, you can potentially make a self-sustaining loop. Even when there’s no sunlight, you can use stored energy in the form of chemical fuel. You can put the hydrogen and oxygen products in separate tanks and incorporate another module like a fuel cell to turn those fuels back into electricity.
Jun Lou, Professor of Materials Science and Nanoengineering, Rice University
The scientists stated that they will continue to enhance the method of encapsulation as well as the solar cells to increase the efficiency of the modules.
The co-authors of the study are former Rice visiting students Xiao Han, who is now an associate professor at Northwestern Polytechnical University in Xi’an, China, and Yunxiu Qiu, a graduate student at Washington University in St. Louis; Rice graduate students Qiyi Fang and Boyu Zhang; Rice alumnus Weipeng Wang, now an assistant professor at Tsinghua University.
The study further involved Rice postdoctoral researcher Jing Zhang; and Pulickel Ajayan, chair of Rice’s Department of Materials Science and Nanoengineering, the Benjamin M. and Mary Greenwood Anderson Professor in Engineering and a professor of chemistry.
The study was financially supported by the Peter M. and Ruth L. Nicholas Postdoctoral Fellowship in Nanotechnology from Rice’s Smalley-Curl Institute, the Welch Foundation, the National Science Foundation-backed Nanosystems Engineering Research Center for Nanotechnology-Enabled Water Treatment, and Fundamental Research Funds for the Central Universities, China.
Journal Reference:
Liang, J., et al (2020) A Low-Cost and High-Efficiency Integrated Device toward Solar-Driven Water Splitting. ACS Nano. doi.org/10.1021/acsnano.9b09053.