Jun 28 2016
Researchers at KTH have found a way to produce hydrogen on a large scale. Discovering a more efficient way to divide water without the aid of rare metals has led the researchers to the finding.
 The research team led by KTH Professor Licheng Sun is one of many worldwide searching for cheaper alternatives to precious metals for large-scale water splitting. (CREDIT: KTH Royal Institute of Technology)
The research team led by KTH Professor Licheng Sun is one of many worldwide searching for cheaper alternatives to precious metals for large-scale water splitting. (CREDIT: KTH Royal Institute of Technology)
Discovering an efficient, reliable, cheap method to extract hydrogen from water can make a hydrogen-fuel economy a reality.
A group of scientists from the KTH Royal Institute of Technology, located in Stockholm, have informed that their finding has overcome a major hurdle in utilizing the renewable energy source.
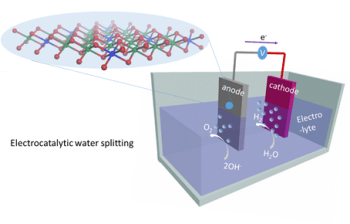
Electrochemical oxidation, more commonly known as “water splitting,” requires the use of costly, precious metal as catalysts. Due to this, a number of research teams across the world are trying to come up with alternative means to substitute them. A group of researchers, headed by KTH Professor Licheng Sun is one such team. Licheng Sun, had tried to create molecular catalysts for water oxidation in the past and succeeded. Information about Sun’s study was published in Nature Chemistry in 2012.
The latest discovery of the team, which has been presented in Nature Communications, reveals that a new earth-abundant material can be used as a catalyst in the process of water-splitting. Using this element can improve the cost-efficiency of large-scale production of hydrogen fuels.
The new alternative material, which contains vanadium and nickel, is a monolayered double hydroxide, says Researcher Ke Fan. This material is an advanced electrocatalyst that can be used for water oxidation. In addition to being efficient and cheap, the nickel-vanadium monolayer also performs better than other non-precious material based electrocatalysts, according to Fan. The material is also a cheaper alternative to catalysts that use expensive, precious metals like ruthenium oxide (RuO2) or iridium oxide (IrO2).
This is the first time that the metal, vanadium, has been used to dope nickel hydroxide to form a water oxidation catalyst, and it works very well — even beyond our expectations. No doubt this material can greatly expand the scope of non-precious metal elements of electrocatalysts, and it opens new areas for water splitting.
Fan
Sun’s catalyst paves way for production of hydrogen fuel in a large scale.
The layered structure of the material contains a monolayer nickel-vanadium oxygen polyhedron that is connected with a thickness less than 1 nanometer, says researcher Hong Chen.
This monolayer feature not only increases the active surface area, but also enhances the electron transfer within the material.
Chen
Sun expects the research to “open a new area of low-cost water oxidation catalysts, featuring stability and efficiencies that equal or even surpass some of today’s best catalysts including RuO2 and IrO2.”